Category: Parkinson's Disease: Neuroimaging
Objective: To investigate free-water (FW) changes in Parkinson’s disease (PD), Multiple system atrophy (MSA) and progressive supranuclear palsy (PSP).
Background: Various parkinsonian syndrome includes PD, MSA and PSP, which show overlapping clinical features may be difficult to diagnose clinically especially in early stages. Several studies had illustrated elevated FW in substantia nigra(SN) and basal ganglia(BG) regions in Parkinsonian disorders with a potential to reliably discriminate them with FW making FW measures a candidate biomarker[1,2]. To investigate FW potential, we evaluated the complete WM and deep gray matter structures.
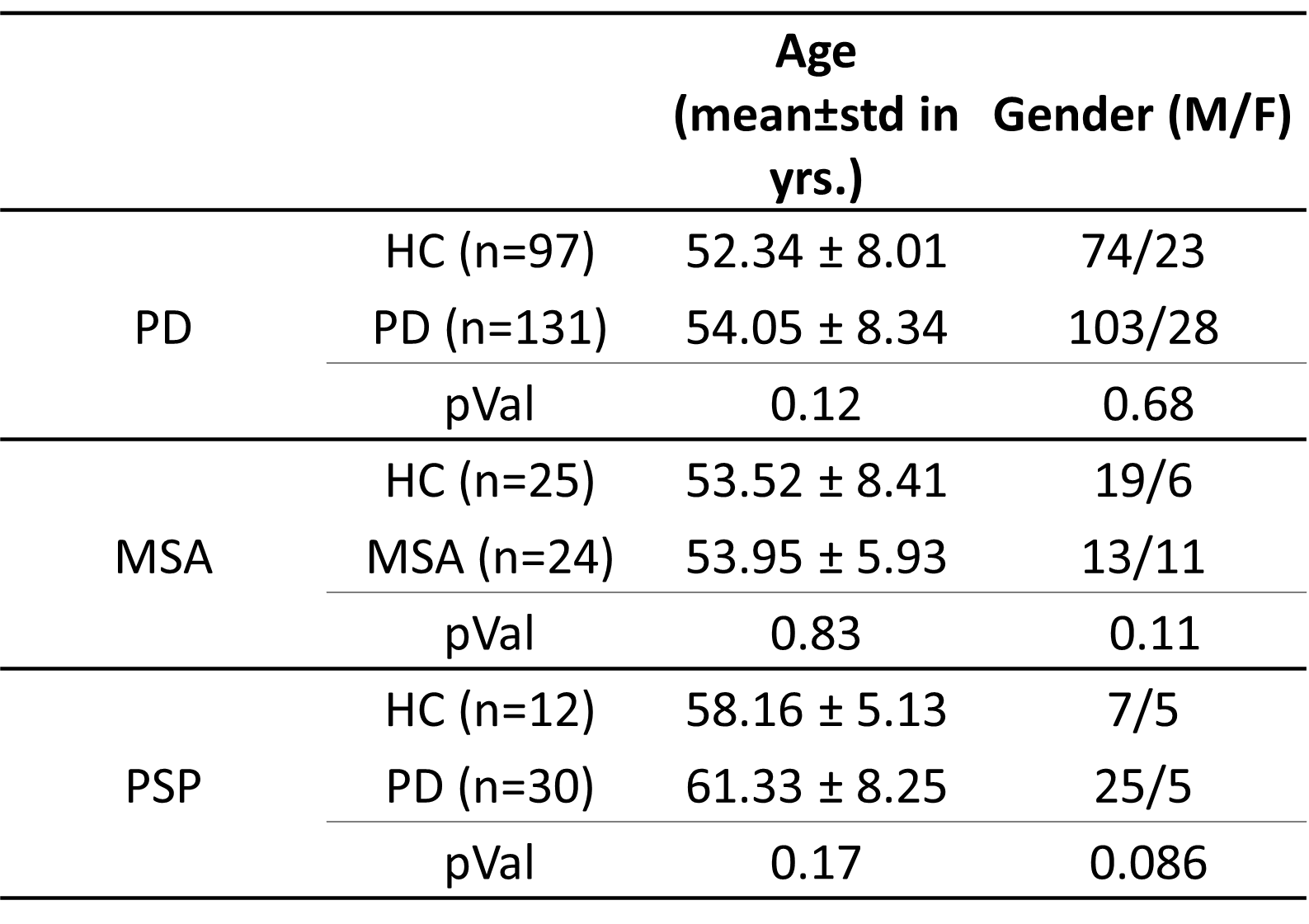
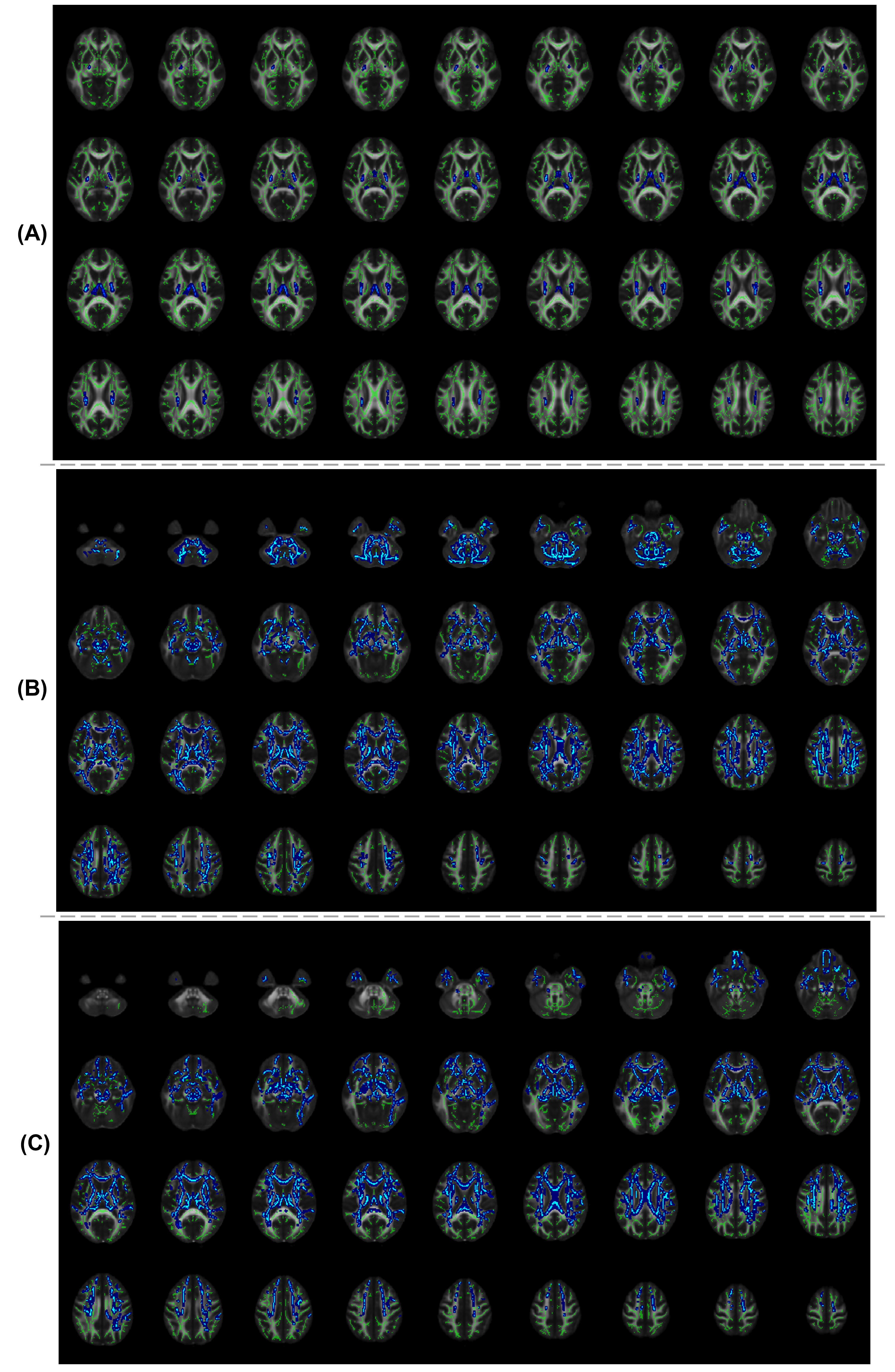
Method: This study includes three parkinsonian datasets and healthy controls shown in table 1. All participants underwent diffusion MRI(dMRI) sequence[3,4,5] on 3T Philips Achieva. The dMRI images were preprocessed using the FSL pipeline[6,7] followed by the FW estimation with a minimization process that fits bi-tensor model[8] to quantify fractional volume of FW for each voxel using Dipy[9]. Tract-based spatial statistics(TBSS)[10] was then employed on the FW maps to analyze the changes in the WM. For gray matter(GM), the mean value of FW extracted for BG regions and SN parscompacta(SNc) atlas defined on neuromelanin MRI[11], which were analyzed using multivariate analysis of covariance test with age-gender as covariates followed by the FDR correction for multiple comparisons.
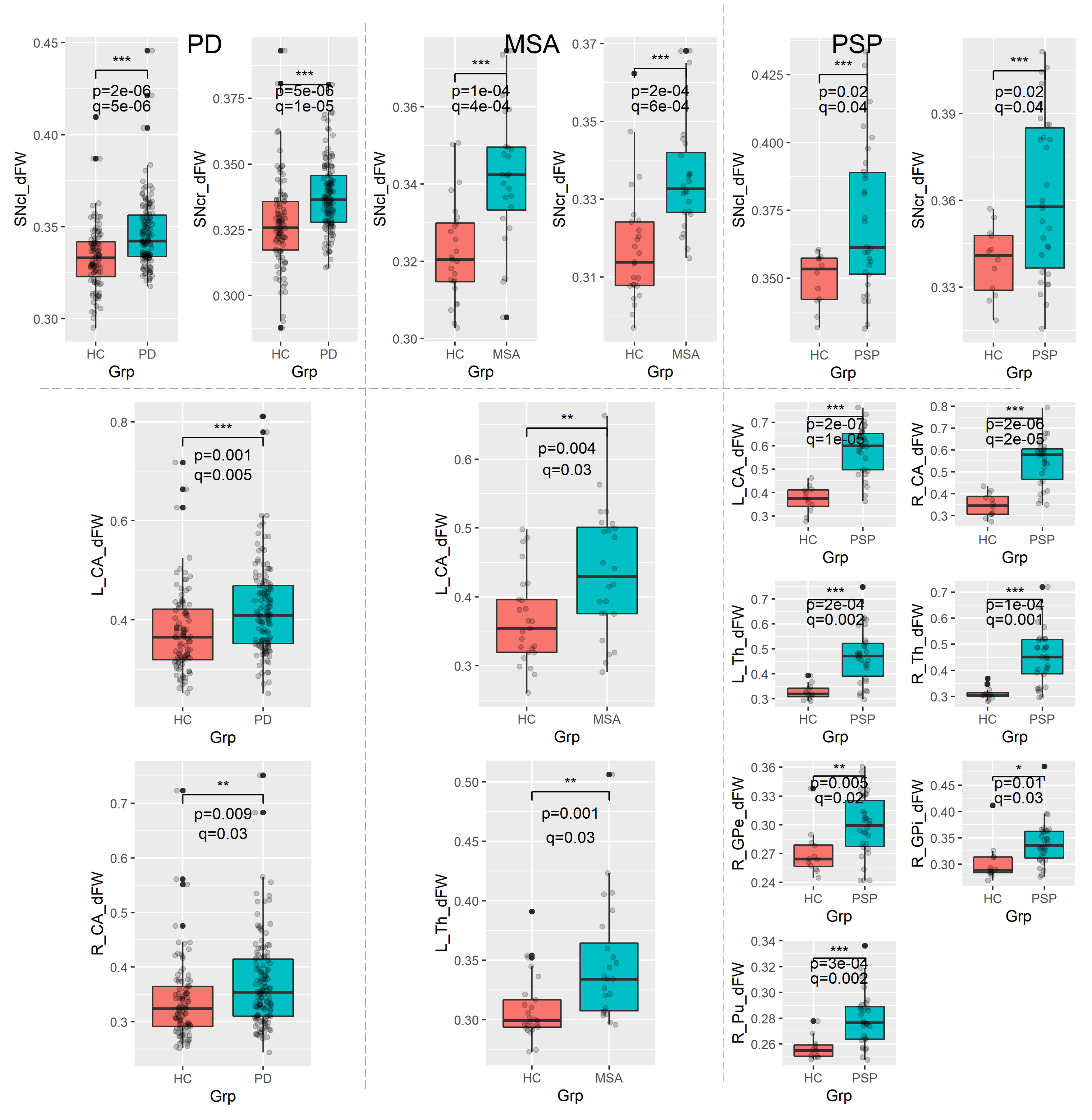
Results: FW was significantly higher in bilateral corticospinal tract(CST), left superior longitudinal fasciculus and left hemisphere forceps major tract in PD patients. While in MSA and PSP, FW was significantly higher in the bilateral CST, anterior thalamic radiation, inferior fronto-occipital fasciculus, uncinate fasciculus and forceps minor tracks compared to HC. Higher spatial extent of significant increase in FW was observed in MSA and PSP as compared to the PD. Increased FW in SNc was seen in all three conditions. FW also showed significant increase at bilateral caudate nucleus(CA) in patients with PD; left-hemispheric thalamus(Th) and CA in MSA and bilateral CA, Th and right-hemispheric global pallidum and putamen in PSP.
Conclusion: Idiopathic PD and atypical parkinsonian syndromes show different patterns and extent of FW increase in various GM and WM structures. SNc shows increased FW in all three disorders. FW can be considered a potential candidate for delineating these disorders.
References: 1. Planetta, P.J., Ofori, E., Pasternak, O., Burciu, R.G., Shukla, P., DeSimone, J.C., Okun, M.S., McFarland, N.R. and Vaillancourt, D.E., 2016. Free-water imaging in Parkinson’s disease and atypical parkinsonism. Brain, 139(2), pp.495-508
2. Andica, C., Kamagata, K., Hatano, T., Saito, A., Uchida, W., Ogawa, T., Takeshige-Amano, H., Zalesky, A., Wada, A., Suzuki, M. and Hagiwara, A., 2019. Free-water imaging in white and gray matter in Parkinson’s disease. Cells, 8(8), p.839.
3. Shah, A., Lenka, A., Saini, J., Wagle, S., Naduthota, R.M., Yadav, R., Pal, P.K. and Ingalhalikar, M., 2017. Altered brain wiring in Parkinson’s disease: a structural connectome-based analysis. Brain connectivity, 7(6), pp.347-356
4. Shah, A., Prasad, S., Rastogi, B., Dash, S., Saini, J., Pal, P.K. and Ingalhalikar, M., 2019. Altered structural connectivity of the motor subnetwork in multiple system atrophy with cerebellar features. European Radiology, 29(6), pp.2783-2791.
5. Prasad, S., Rajan, A., Pasha, S.A., Mangalore, S., Saini, J., Ingalhalikar, M. and Pal, P.K., 2021. Abnormal structural connectivity in progressive supranuclear palsy—Richardson syndrome. Acta Neurologica Scandinavica, 143(4), pp.430-440.
6. Smith, S.M., Jenkinson, M., Woolrich, M.W., Beckmann, C.F., Behrens, T.E., Johansen-Berg, H., Bannister, P.R., De Luca, M., Drobnjak, I., Flitney, D.E. and Niazy, R.K., 2004. Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage, 23, pp.S208-S219.
7. Andersson, J.L. and Sotiropoulos, S.N., 2016. An integrated approach to correction for off-resonance effects and subject movement in diffusion MR imaging. Neuroimage, 125, pp.1063-1078.
8. Pasternak, O. , Sochen, N. , Gur, Y. , Intrator, N. and Assaf, Y. (2009), Free water elimination and mapping from diffusion MRI. Magn. Reson. Med., 62: 717-730.
9. Henriques, R.N., Rokem, A., Garyfallidis, E., St-Jean, S., Peterson, E.T. and Correia, M.M., 2017. [Re] Optimization of a free water elimination two-compartment model for diffusion tensor imaging. bioRxiv, p.108795.
10. S.M. Smith, M. Jenkinson, H. Johansen-Berg, D. Rueckert, T.E. Nichols, C.E. Mackay, K.E. Watkins, O. Ciccarelli, M.Z. Cader, P.M. Matthews, and T.E.J. Behrens. Tract-based spatial statistics: Voxelwise analysis of multi-subject diffusion data. NeuroImage, 31:1487-1505, 2006
11. Safai, A., Prasad, S., Chougule, T., Saini, J., Pal, P.K. and Ingalhalikar, M., 2020. Microstructural abnormalities of substantia nigra in Parkinson’s disease: a neuromelanin sensitive MRI atlas based study. Human brain mapping, 41(5), pp.1323-1333.
To cite this abstract in AMA style:
AKS. Shah, JS. Saini, MI. Ingalhalikar, SP. Prasad, PKP. Pal. Diffusion-weighted imaging based free-water investigation in Parkinsonian disorders [abstract]. Mov Disord. 2022; 37 (suppl 2). https://www.mdsabstracts.org/abstract/diffusion-weighted-imaging-based-free-water-investigation-in-parkinsonian-disorders/. Accessed April 18, 2025.« Back to 2022 International Congress
MDS Abstracts - https://www.mdsabstracts.org/abstract/diffusion-weighted-imaging-based-free-water-investigation-in-parkinsonian-disorders/