Objective: To evaluate the potential added benefit of opicapone (OPC), compared with placebo (PLC) and entacapone (ENT), in catechol-O-methyltransferase inhibitor (COMTi)-naïve Parkinson’s disease (PD) patients recently diagnosed with motor fluctuations (MF).
Background: OPC, a once-daily COMTi, was shown to be effective for end-of-dose MF in PD patients in two large multinational trials (BIPARK-I and II) [1, 2].
Method: PLC, OPC 50 mg and ENT data from COMTi-naïve patients in BIPARK-I [1] were analysed. Primary efficacy endpoint was change from baseline in OFF-time based on patient diaries. Scale-based secondary efficacy endpoints included changes from baseline in the Unified Parkinson’s Disease Rating Scale (UPDRS), Parkinson’s Disease Sleep Scale (PDSS), 39-item Parkinson’s Disease Questionnaire (PDQ-39), Non-Motor Symptoms Scale (NMSS), and Clinician and Patient Global Impression of Change (CGI-C and PGI-C). Safety was assessed by evaluating the incidence of treatment-emergent adverse events (TEAEs) considered at least possibly drug-related. Recently diagnosed patients with MF were ‘recent fluctuators’, defined as those having an onset of MF </=1 year of study baseline.
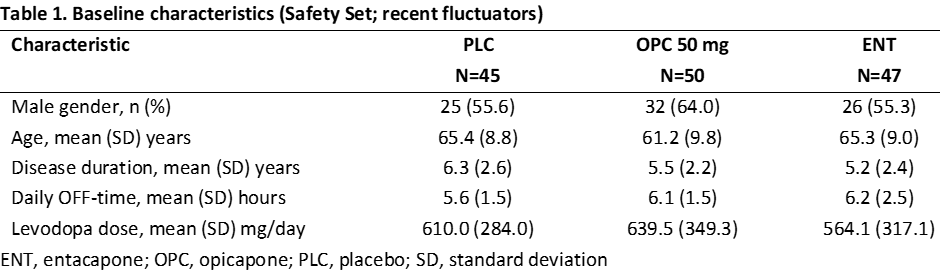
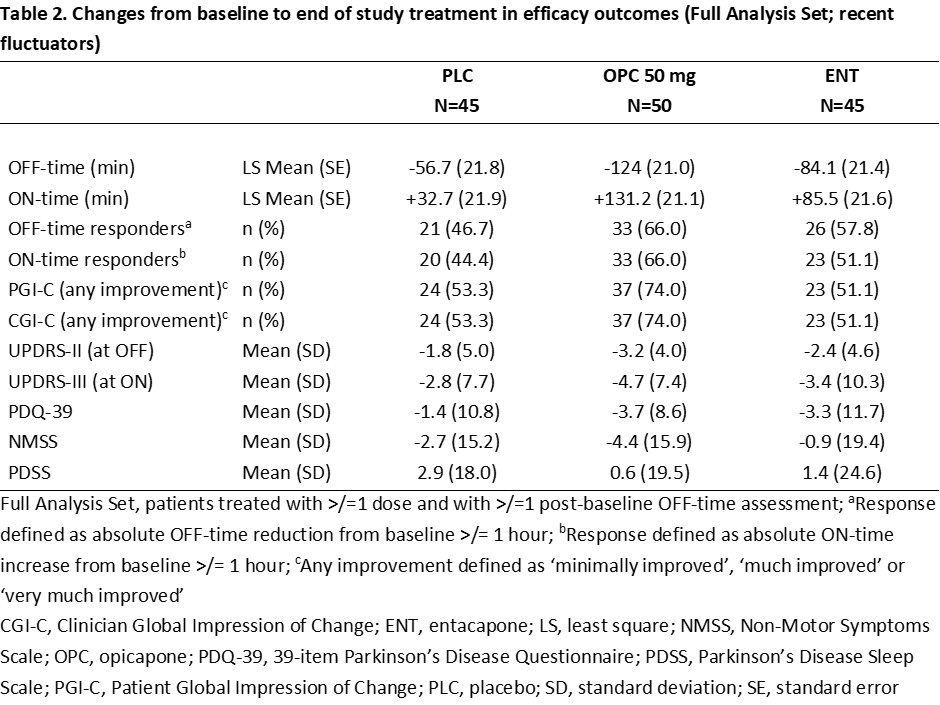
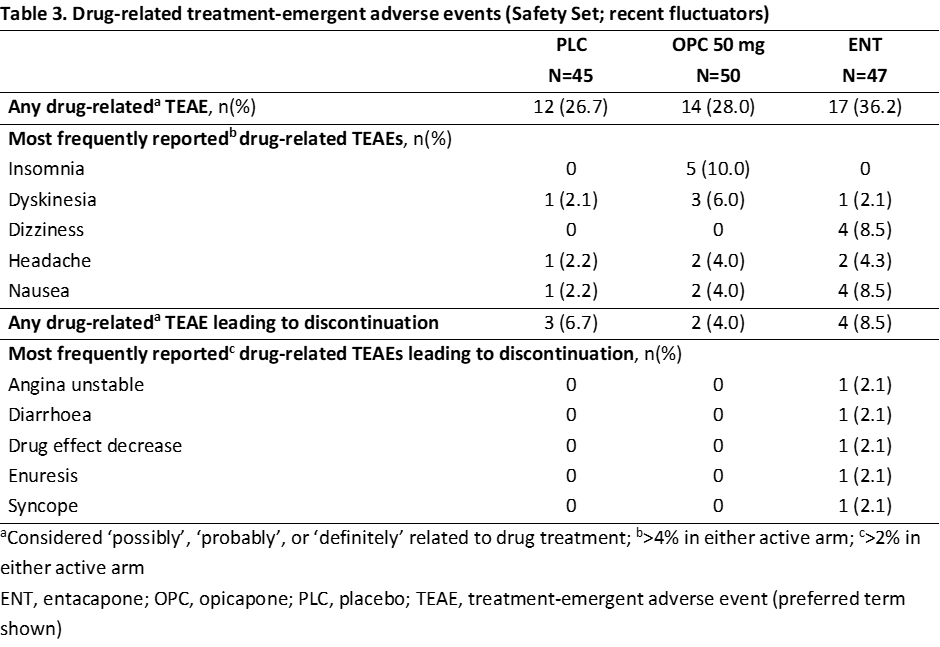
Results: Overall, 142 COMTi-naïve patients who were recent fluctuators were randomised to PLC (n=45), OPC 50 mg (n=50) or ENT (n=47) and included in this post-hoc analysis (Table 1). Treatment with OPC 50 mg resulted in greater OFF-time reduction and ON-time increase versus both PLC and ENT (Table 2). Higher proportions of OPC 50 mg-treated patients versus both PLC and ENT were considered OFF-/ON-time responders and exhibited improvements, as assessed by CGI-C and PGI-C (Table 2). Additionally, OPC 50 mg resulted in greater numerical improvements from baseline in UPDRS, NMSS, and PDQ-39 scores than both PLC and ENT (Table 2). The overall incidences of drug-related TEAEs and drug-related TEAEs leading to discontinuation were lower for OPC 50 mg versus ENT (Table 3).
Conclusion: Overall OPC 50 mg demonstrated added benefit as a first adjunctive COMTi, in comparison with PLC and ENT, in levodopa-treated PD patients recently diagnosed with MF.
References: 1. Ferreira JJ, et al. Lancet Neurol. 2016;15:154–65. 2. Lees A, et al. JAMA Neurol. 2017;74:197–206.
To cite this abstract in AMA style:
A. Lees, J. Ferreira, W. Poewe, H. Gama, D. Magalhães, J.F Rocha, P. Soares-da-Silva. Efficacy and safety/tolerability of opicapone in catechol-O-methyltransferase inhibitor-naïve Parkinson’s disease patients recently diagnosed with motor fluctuations [abstract]. Mov Disord. 2020; 35 (suppl 1). https://www.mdsabstracts.org/abstract/efficacy-and-safety-tolerability-of-opicapone-in-catechol-o-methyltransferase-inhibitor-naive-parkinsons-disease-patients-recently-diagnosed-with-motor-fluctuations/. Accessed April 2, 2025.« Back to MDS Virtual Congress 2020
MDS Abstracts - https://www.mdsabstracts.org/abstract/efficacy-and-safety-tolerability-of-opicapone-in-catechol-o-methyltransferase-inhibitor-naive-parkinsons-disease-patients-recently-diagnosed-with-motor-fluctuations/